 Revenue: Total revenue was $14.6 million for the quarter ended June 30, 2021, as compared to $3.6 million for the quarter ended June 30, 2020. We expect cash and cash equivalents to be sufficient to fund operations into the second half of 2023. 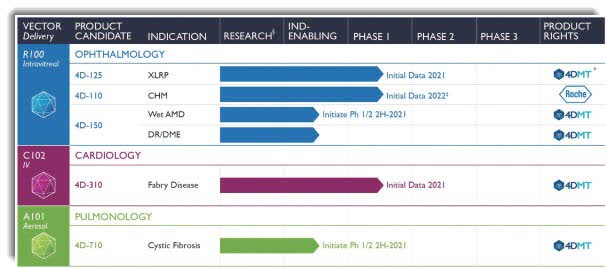
Initiation of a clinical trial with 4D-710 in cystic fibrosis lung disease expected in the fourth quarter of 2021įinancial Results for the Second Quarter Ended June 30, 2021Ĭash and Cash Equivalents: Cash and cash equivalents were $243.7 million as of June 30, 2021.Initiation of a clinical trial with 4D-150 in wet AMD expected in the fourth quarter of 2021.Initial clinical activity data from the Phase 1 clinical trial of 4D-110 in choroideremia expected in the fourth quarter of 2021.Initial clinical activity data from the Phase 1/2 clinical trial of 4D-125 in XLRP expected in the fourth quarter of 2021. 
Initial clinical data from the Phase 1/2 clinical trial of 4D-310 in Fabry disease expected in the fourth quarter of 2021.We also plan to submit to the FDA safety and efficacy data from the 4D-110 Phase 1 clinical trial along with a new 4D-110 clinical study protocol in the fourth quarter of 2021. We expect to report initial clinical activity data from the Phase 1 clinical trial of 4D-110 in choroideremia and the Phase 1/2 clinical trial of 4D-125 in XLRP in the fourth quarter of 2021. Updated our rare disease ophthalmologic portfolio: reported positive initial safety data from the Phase 1 clinical trial of 4D-110 in choroideremia and the Phase 1/2 clinical trial of 4D-125 in X-linked retinitis pigmentosa (XLRP) and regained global rights to 4D-110.in Organization Development and Clinical Psychology. Greiner has served as head of human resources for 25 years at organizations including California Pacific Medical Center, the Palo Alto Medical Foundation at Sutter Health, The Institute on Aging, and Bank of America. Zimmermann brings over 27 years of leadership experience in life sciences corporate and business development, including from her prior roles at Johnson & Johnson Innovation and Novartis Pharmaceuticals. Appointed Carolyne Zimmermann as Chief Business Officer.The expanded facility will enable commercial-scale manufacturing of our clinical product candidates and expansion of our analytical development capabilities, including laboratories dedicated for developing potency assays. 
Initiated the expansion of current Good Manufacturing Practices (cGMP) compliant manufacturing facilities and analytical laboratories at 4DMT headquarters in Emeryville, CA to support manufacturing and analytical testing of current and future product candidates.We remain on track to announce clinical data from each of our three ongoing clinical programs and to initiate clinical development with product candidates in wet AMD and cystic fibrosis by the end of this year.” In addition, we enhanced our leadership team with the additions of Carolyne Zimmermann as Chief Business Officer and Nadine Greiner, Ph.D., as Chief Human Resources Officer. This achievement underscores the promise of our Therapeutic Vector Evolution platform, which empowers us to invent targeted and evolved vectors that package efficiently during our proprietary manufacturing process. Notably, we believe we are the first AAV gene therapy company to successfully complete cGMP manufacturing of primate-evolved targeted vectors, and we achieved this milestone with three different clinical product candidates. “Importantly, we initiated the expansion of our cGMP manufacturing facilities to support commercial-scale production of our clinical product candidates. “Harnessing the power of directed evolution to develop targeted gene therapies is central to 4DMT, and this past quarter we continued to make substantial progress towards our goal,” said David Kirn, M.D., Co-founder and Chief Executive Officer of 4DMT. 12, 2021 (GLOBE NEWSWIRE) - 4D Molecular Therapeutics (Nasdaq: FDMT), a clinical-stage gene therapy company harnessing the power of directed evolution for targeted gene therapies, announced financial results for the second quarter of 2021, and provided operational highlights. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
